Contact Line Pinning
The residue left behind from evaporating raindrops can be an unwelcome sight on your newly-washed car. But the same evaporative process can be beneficial, for example, in inkjet printing where drop residue becomes part of a printed image or text.
The residue from evaporating liquid drops can appear in unexpected ways. Coffee ring stains are a well-known example, in which coffee residue collects on the outer edge of a drop, leaving a thin circular ring stain. This phenomenon is the result of some interesting fluid dynamics processes. In order for the coffee ring stain to form, the liquid must have a pinned contact line on the solid surface on which it has been deposited. A pinned contact line means that the outer edge of the liquid drop, where it intersects the solid substrate, remains stationary as the drop evaporates. Evaporation is caused by heat in the substrate and is greatest at the thin outer edge of the drop. Surface tension causes liquid to flow towards the edge to replace the liquid lost to evaporation. This, in turn, carries more solute to the edge, resulting in a higher concentration of solute residue that forms the eventual coffee ring stain after all of the liquid evaporates.
Modeling Approach
A new contact line pinning model has been developed for the latest update of FLOW-3D v12.0, expanding the software’s capabilities to a wider range of surface-tension-dominated applications. Interface pinning and de-pinning surface properties play an important role in inkjet printing, coating and spray cooling. Surface engineering with respect to its wetting properties may be used to control the movement of liquid samples in microfluidic devices.
The main feature of the model is providing the means of pinning the edge of the drop to a fixed position. Geometry component and subcomponent surfaces can be assigned the ‘pinning’ attribute. The fluid contact line becomes pinned where it first comes in contact with such a surface. The pinning is enforced by keeping the front velocity equal to zero. Fluid can move past the contact point only by rolling over it, not by moving the contact line along the surface.
Coffee Ring Stain Validation
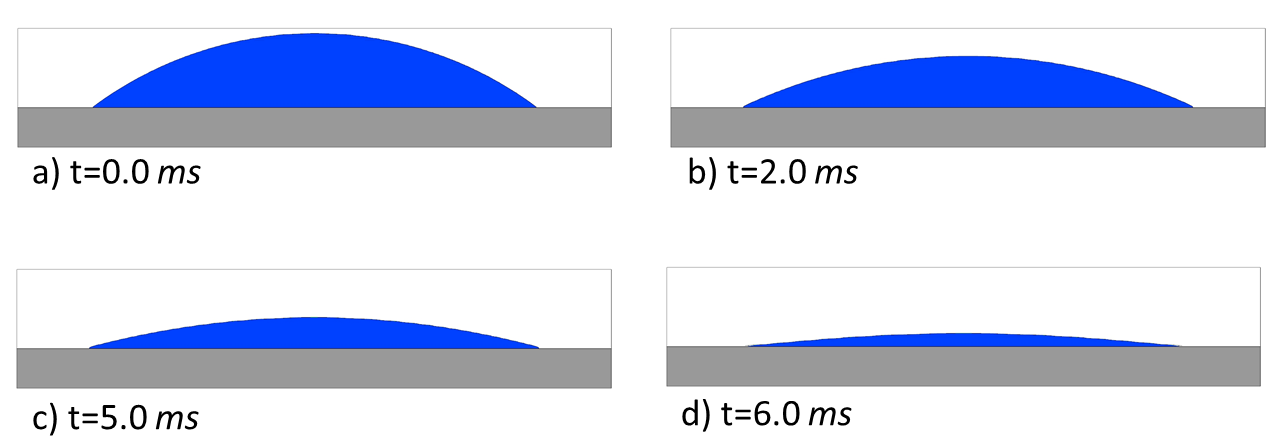
Figure 1 shows results for a circular droplet placed on a horizontal flat surface. The surface is kept at a constant temperature of 30℃. The initial fluid temperature is 20℃, and the surrounding void is at the constant temperature of 20℃. Fluid represents a typical ink with the density of 0.967 g/cm3, viscosity 0.02022 poise, specific heat 1.645e+07 cm2/s/K, thermal conductivity 1.2964e+4 g cm/s3/K, and the surface tension coefficient of 33.15 g/cm2.
The initial radius of curvature of the droplet’s surface is 7.5e-03 cm, its footprint is a circle of radius 4.5e-03 cm, and the apparent initial contact angle is 37.87 degrees, as shown in Figure 1a. The specified static contact angle is 0 degrees.
The constant pressure phase change model is activated. The partial pressure of vapor in the void is zero, and the phase change accommodation coefficient Rsize=0.01.
The residue model is also turned on to capture the formation of the solid residue on the substrate when the ink dries. The concentration of pigment dissolved in the fluid is represented by a transportable scalar at the initial concentration of 0.01 g/cm3 and the maximum concentration of rmax=1.1625 g/cm3. The dissolved pigment enhances the fluid net viscosity based on mass averaging, at the rate of 0.05 poise per unit mass of the pigment.
The process is modeled in an axisymmetric cylindrical mesh spanning 3.0 degrees and one cell in the azimuthal direction; dx=6e-05 cm, dz=4e-05 cm.
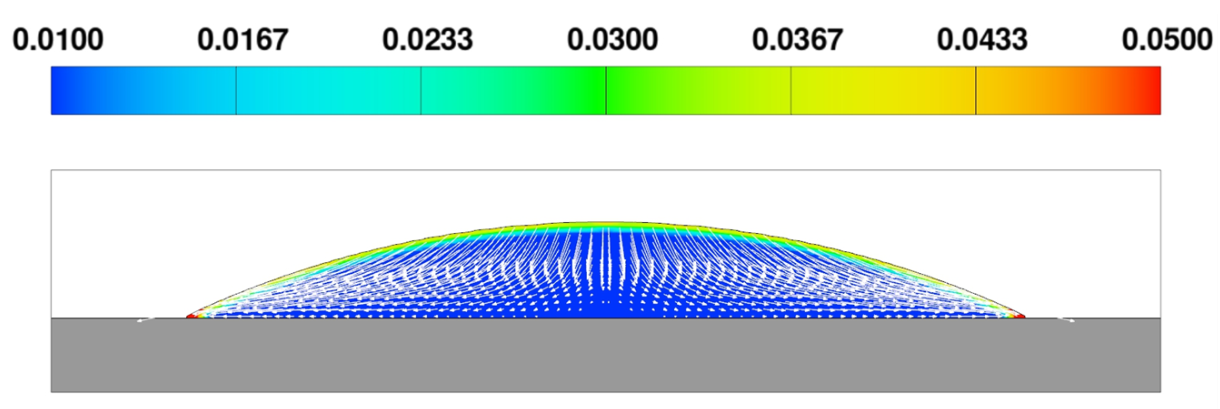
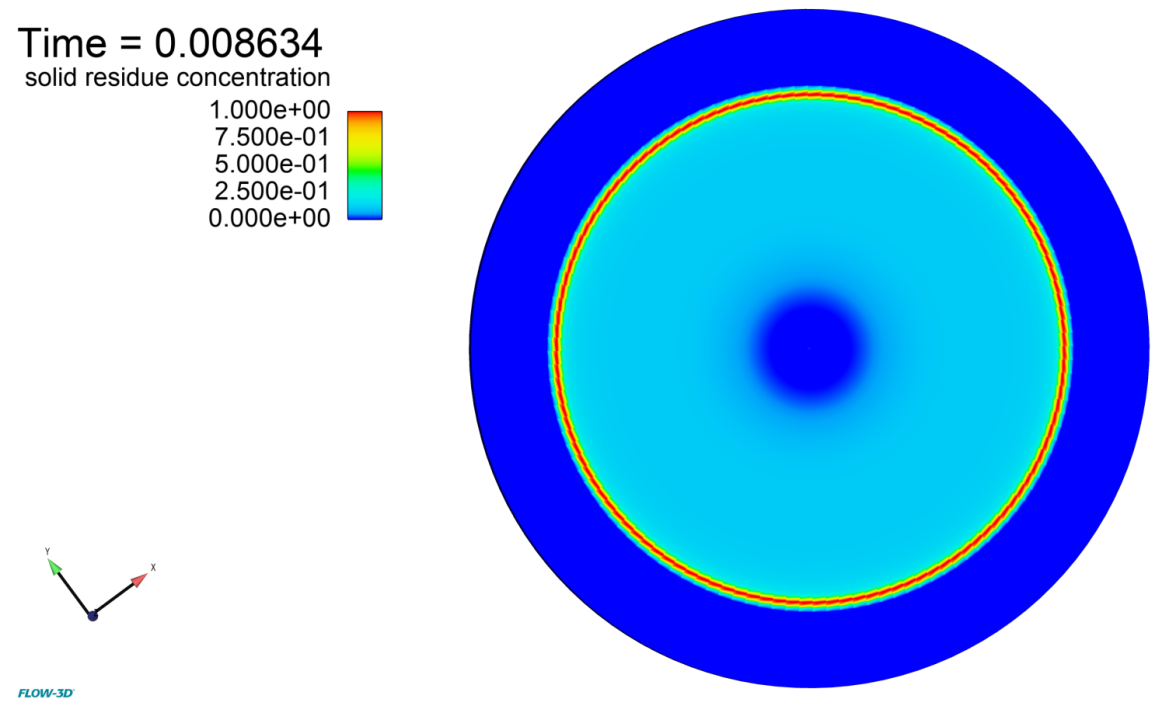
Figure 1 shows that, as fluid evaporates, its contact line stays pinned. The static contact angle condition of 0 degrees results in a pressure gradient directed towards the center of the droplet, which in turn creates a flow in the direction of the contact line. The concentration of the dissolved pigment increases near the free surface due to evaporation, and the flow redistributes it further towards the contact line (Figure 2). As the liquid continues to evaporate, the concentration of the pigment in the remaining liquid increases. When it reaches the maximum rmax, the excess pigment is converted to solid residue.
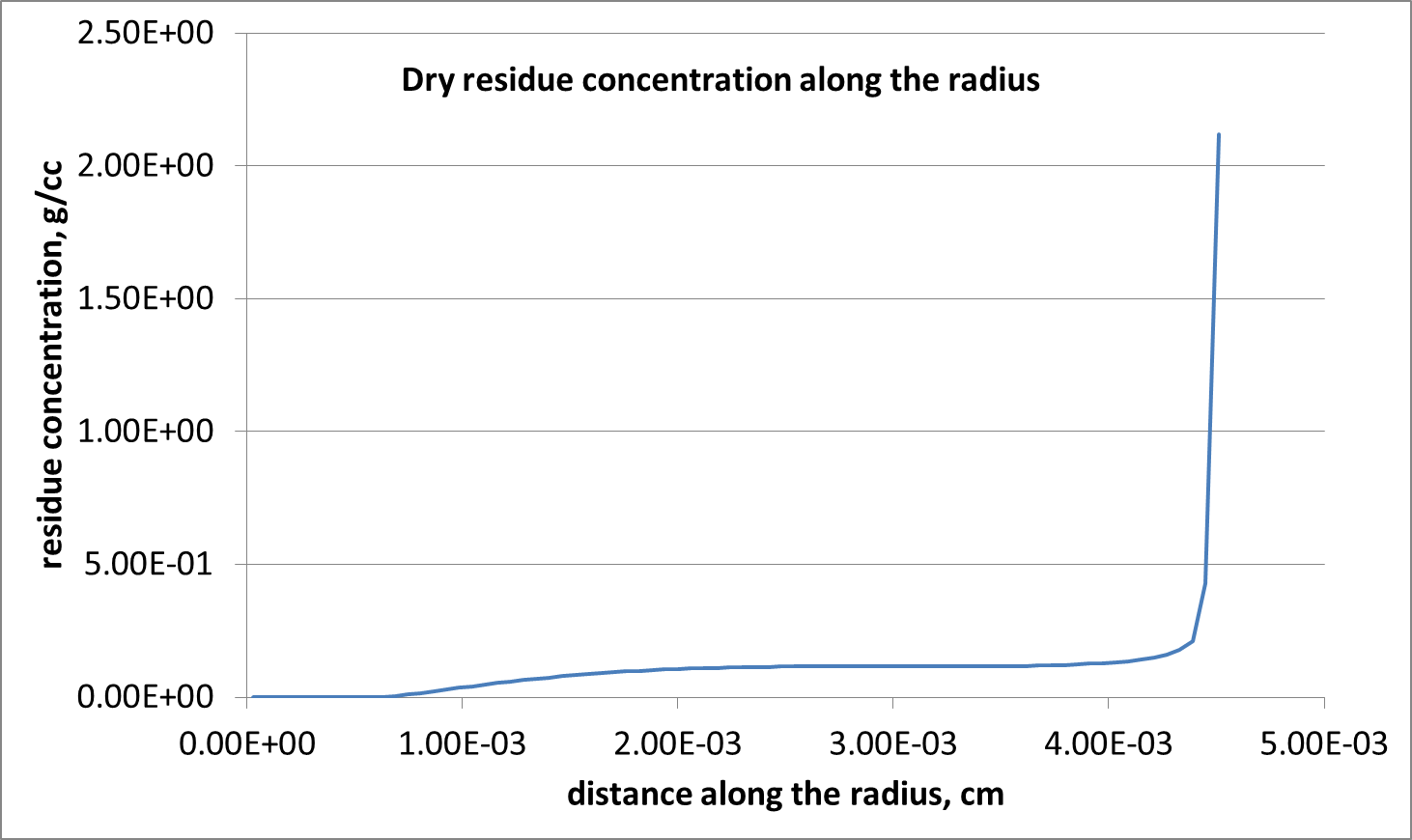
Fluid near the contact line dries first, leaving the solid residue behind. Due to the higher concentration of the pigment in the fluid at that area, a characteristic ‘coffee ring’ pattern of the residue is created on the surface of the substrate (Figures 3 and 4). The total mass of the pigment – dissolved plus dry residue – is conserved to within 0.025% of the initial mass.
Droplet on a Wall Validation
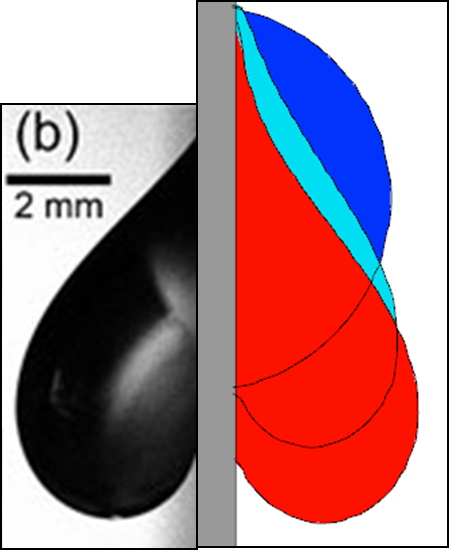
The second example of a contact line pinning application shows the behavior of a drop of liquid aluminum pinned on a vertical wall. The fluid density is 2.7 g/cm3, surface tension coefficient 200 g/cm2, and viscosity 0.27 poise. The static contact angle is 0 degrees.
A droplet of radius 0.5 cm is placed on the vertical wall so that the initial apparent contact angle is 90 degrees (Figure 5). The magnitude of gravity, 7e+06 cm/s2, is artificially enhanced to overcome the restoring action of the surface tension forces and produce a noticeable deformation of the droplet. The results are shown in Figure 5, including a qualitative comparison with an experimental result for a similar-sized droplet.
Summary
FLOW-3D‘s contact line pinning model extends the surface tension and wall adhesion capabilities in order to model complex interactions at engineered surfaces. More examples and additional references where contact line pinning is finding practical applications can be found here.