
Applying computational fluid dynamics to the field of biotechnology is a relatively new practice and has great potential to improve the way many medical devices are used or implemented. FLOW-3D is a robust simulation tool with an extensive range of integrated capabilities, including free-surface/confined flows, variable density, phase change, moving objects, and mechanical and thermal stress analysis.
Active Mixing
Active mixers make use of a mechanical device to facilitate mixing. In this example a magnetic stir bar (driven by an external rotating magnetic field) is used to induce mixing. This simulation uses the Moving Objects model to simulate the magnetic stir bar. The magnetic interaction of the bar is not included but instead a rotational speed (RPM) of the bar is specified directly as a prescribed motion. In this case, there is no density variation, however, FLOW-3D can easily simulate multiple density components, i.e., miscible or immiscible fluids. The separation of immiscible fluids in FLOW-3D is based on a drift relationship.
This simulation demonstrates an active micro mixing process. The flow is from left to right. This is a one-fluid simulation with the bottom half (red) being seeded with passive scalars, or tracers, and therefore there is no density gradient.
Chemical Reactions
Acid Base Neutralization: HCl + NaOH –> NaCl
FLOW-3D has a chemical reaction model where multiple components can create a new concentration. For instance, in this example, 20 liters of 1M Hydrochloric Acid (HCl is mixed with 2 milliliters of Sodium Hydroxide (NaOH) to make NaCL, or salt.
The mixing is accelerated with a rotating stirrer at 4 pi radians/sec (120 rpm). The images on the left display the concentrations of the acid (HCl), the base (NaOH) and the product (NaCl), respectively, while the image on the right is a 3D view of the base concentration. The stirrer is rotating at 4 radians per second.
Mixing & Chemical Reactions: Acid Base Neutralization: With and Without Stirrer
The simulation shown below with an active stirrer was compared against passive mixing (or diffusion only). Run passively, the reaction takes 437 seconds to reach a pH of 7. The stirrer accelerates the process to where it only takes 20 seconds at a rotation rate of 4 pi radians per second. The Moving Objects Model was used to simulate the rotating mixer and the chemical reaction model was used to simulate the reactions. A time-dependent mass/momentum source introduces the base substance into the chamber.
With Stirrer
Without Stirrer
Drug Delivery
Shown here is a transient, free-surface simulation of an injection needle modeled using cylindrical coordinates in conjunction with FLOW-3D’s Moving Objects Model. The needle itself is a few hundred micrometers wide while the syringe section is a few millimeters. A time-dependent prescribed velocity is applied to the plunger to generate the motion. In this application it is important to understand the injection force required on the plunger while squeezing out a shear thinning fluid. FLOW-3D‘s non-Newtonian viscosity model is used to account for these effects. FLOW-3D can include other viscous effects such as thixotropic fluids, temperature-dependent viscosity, a Carreau model or power law. Strain and shear stress are calculated to determine whether proteins and/or cells are damaged.
FLOW-3D is used to predict transient injection forces on a plunger and strain rate for various solutions of shear thinning medical fluid. Medical fluids contain essential components that undergo high strain through shear thinning at the syringe-needle interface. FLOW-3D can be used to model various syringe-needle geometries and fluids to analyze the strain experienced by fluid in passage through the needle as well as the force generated on the plunger for automated injections.
Drug Particles
Nasal drug delivery system, a promising non-invasive means of administrating drugs, poses numerous design challenges. If the spray particles are too large they tend to deposit in the anterior portion of the nasal cavity which reduces the effectiveness of the drug. If the particles are too small, a majority of the particles could pass directly to the lungs wasting most of the dose. For some drugs it is desirable to target a specific region of the nasal cavity like the olfactory region, which has a potential of delivering drugs directly to the brain. Designing a successful nasal drug delivery system requires optimizing various factors such as drug particle size, particle speed, spray angle, and nozzle insertion depth. Using FLOW-3D, the effects of the various design parameters on the system can be easily studied by running a series of simulations.
This simulation shows the trajectory of spray particles through the nasal cavity. Particles are colored by size, where red indicates the largest particles and blue the smallest. The simulation shows that the smaller the particles are, the deeper they penetrate into the nasal cavity. The smallest particles then exit the nasal cavity and go further into the respiratory system.
Microdispensers
This example shows a simulation of a retracting fluid dispenser. The main concern here is the exact volume of fluid dispensed and how much is drawn back into the dispenser. The nozzle is only 1 millimeter in diameter, and at this scale surface tension is important because it dictates the amount of fluid deposited onto the substrate, similar to capillary flow. Using cylindrical coordinates with a specified contact angle between the resin-like fluid, the dispenser nozzle, and the substrate, the model is able to account for these effects. In addition, the Moving Objects Model was used to simulate the dispenser motion. Modeling the complexities of small, transient, free-surface flows along with moving objects can be easily accomplished using FLOW-3D.
Passive Mixing
The mixing of multiple streams of fluids in micro channels/chambers is of great importance in bio-analytical systems. Typically these processes need to occur within short mixing lengths and time scales. Passive micro-mixers make use of the geometry of the micro-channel to induce mixing. Shown here is a design variation where the liquid plus reagent side uses a passive scalar to show the mixing concentration. In this case, the two fluids had nearly identical viscosities but FLOW-3D is capable of modeling different viscosities and densities mixing as well. The video on the right shows a demonstration of a passive micro channel mixing process. The colors represent the concentration of the two different fluids (blue = Pure liquid, red = Liquid + Reagent).
Piezo Driven Pumps
Piezo driven pumps are becoming commonplace in today’s markets and FLOW-3D can easily model the physics involved. In the animation below, a void pressure is set on the back side of the piezoelectric device which is the thin horizontal component. Two valves are included using the Moving Objects Model to control the flow direction as the pump oscillates. An oscillating force is applied to move the membrane. This force can be a point source or distributed. Particles are seeded into the animation to help visualize the flow.
Dissolution with Medical Test Strips
Medical test strips used for rapid diagnostics and determination of relevant biomedical parameters have undergone considerable advances in recent years. Far from the conventional strips with absorbent fleece, new precisely-fabricated microchannels, with defined geometric structures, are now possible via Microsystems engineering. Proper geometrical design of these microstructures enables selective control of the capillary-driven flows. FLOW-3D can be used to optimize the designs of these strips
Regardless of the type of solid, whether it is salt or acetaminophen, penicillin, or sugar, understanding how fast and how much of the solid gets dissolved into the flow is important for several applications areas, especially in medicine. The way in which the solid dissolves could lead to high blood pressure, liver or kidney strain. Also, many pharmaceutical pills are more dense or water-soluble than others and these properties directly affect how the solid interacts with the surrounding medium. All of these parameters are easily varied in FLOW-3D.
FLOW-3D‘s meshing method has solid geometry independence using the FAVOR™ method, and so the area and volume fractions that make up the geometry are simply rewritten at every time step as salt is dissolved from the surface. This makes the simulation very computationally efficient because no mesh refinement is required.
Read more about the Salt Dissolution model in the Modeling Capabilities section.
Agitational Stresses
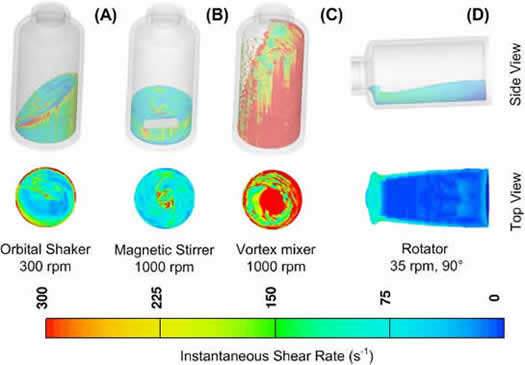
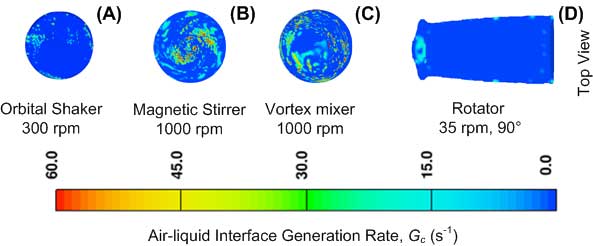
Agitation studies are a common and important part of biopharmaceutical development, yet the fundamental nature of the stresses involved and their impact on protein stability are not fully understood. Characterization of the agitation stress methods is critical to identifying protein degradation mechanisms or specific sensitivities. Stresses caused by shear, interfaces, cavitation, or other fluid and interfacial forces are difficult or impossible to measure with experimental methods. Recently, we conducted CFD simulations with FLOW-3D to model the hydrodynamics of liquid agitated in a 3-mL glass vial with different agitation instruments including a rotator, orbital shaker, magnetic stirrer, and vortex mixer, at varying frequencies to identify and quantify stresses of potential importance to protein stability. The fluid properties of water at 25°C were used for these simulations.

The standard FLOW-3D code was customized so that potentially harmful stresses to proteins such as maximum system shear rate, volume-averaged shear rate, volume-averaged shear rate near air-liquid and solid-liquid interfaces, total shear, area of solid-liquid interface, and air-liquid interface regeneration rate can be numerically calculated and compared as additional outputs of the standard software package. We validated the CFD models by comparing the shape of the free surface of the fluid in the vial between simulation and experiment.
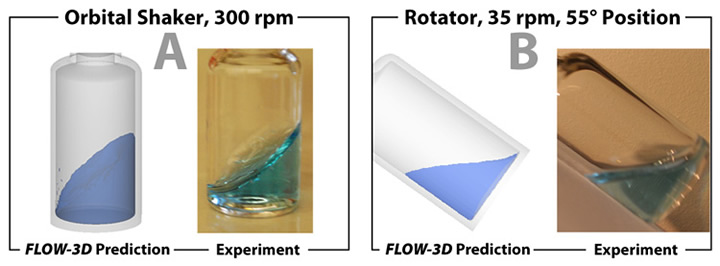
Examples of stresses (shear rate and interface generation rate) and comparisons at the air-liquid and solid-liquid interfaces are shown in Figure 3 and in Figure 4. Overall, the vortex mixer provides the most intense stresses, while the magnetic stirrer presented locally intense shear to the hydrophobic stir bar surface. The rotator provides gentler fluid stresses, but the air-water interfacial area and surface stresses are relatively high given its low rotational frequency. The orbital shaker provides intermediate-level stresses but with the advantage of a large stable platform for consistent vial-to-vial homogeneity.
We found that multiple stresses act concurrently on the liquid in a glass vial in each of the agitation methods described. These stresses varied for different methods and were often found to be strong functions of agitation frequency. We also determined that selecting the proper agitation method with known types and intensities of stresses can facilitate better understanding of the impact on protein degradation mechanisms. We concluded that CFD can be instrumental in characterizing fluid stresses in experimental systems and in validating their relevance to real world conditions.
This article was contributed by Ge Bai, Scientist, MedImmune LLC.
Modeling Capabilities
As a general-purpose CFD software, FLOW-3D has extensive physical and numerical capabilities that have been developed over its 40-year history, such as incompressible free-surface internal and external flow, heat transfer, turbulence, moving and deforming solids, surface tension and phase change. These models allow our customers to successfully use FLOW-3D to solve a wide range of engineering and scientific problems, optimize designs and gain insight into complex processes.
A one-fluid VOF approach, combined with FLOW-3D‘s adiabatic bubble and surface tension models, allows for effective modeling of void regions with a fluid. The solution is fast, robust and accurate, and bypasses the need for more cumbersome 2-fluid VOF approaches.
